Maximum quantity allowed is 999
请选择数量
CAS RN: 603-35-0 | 產品號碼: T0519
產品規格
| Appearance | White powder to lump |
| Purity(Iodometric Titration) | min. 95.0 % |
| Melting point | 80.0 to 82.0 °C |
| Solubility in Toluene | very faint turbidity |
| NMR | confirm to structure |
性質
| 熔點 | 81 °C |
| 沸點 | 260 °C/25 mmHg |
| 水溶性 | Insoluble |
| Degree of solubility in water | 0.3 mg/l 25 °C |
| 溶解性(可溶於) | Chloroform, Benzene, Toluene |
| 溶解性(微溶於) | Alcohol |
GHS
| 圖形表示 |


|
| 信號詞 | Danger |
| 危險性說明 | H302 : Harmful if swallowed. H315 : Causes skin irritation. H319 : Causes serious eye irritation. H372 : Causes damage to organs through prolonged or repeated exposure. H373 : May cause damage to organs through prolonged or repeated exposure. H317 : May cause an allergic skin reaction. H335 : May cause respiratory irritation. H413 : May cause long lasting harmful effects to aquatic life. |
| 防範說明 | P501 : Dispose of contents/ container to an approved waste disposal plant. P273 : Avoid release to the environment. P272 : Contaminated work clothing should not be allowed out of the workplace. P260 : Do not breathe dust/ fume/ gas/ mist/ vapors/ spray. P270 : Do not eat, drink or smoke when using this product. P271 : Use only outdoors or in a well-ventilated area. P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P302 + P352 : IF ON SKIN: Wash with plenty of water. P314 : Get medical advice/ attention if you feel unwell. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P305 + P351 + P338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P362 + P364 : Take off contaminated clothing and wash it before reuse. P333 + P313 : If skin irritation or rash occurs: Get medical advice/ attention. P301 + P312 + P330 : IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. Rinse mouth. P304 + P340 + P312 : IF INHALED: Remove person to fresh air and keep comfortable for breathing. Call a POISON CENTER/doctor if you feel unwell. P403 + P233 : Store in a well-ventilated place. Keep container tightly closed. P405 : Store locked up. |
相關法規
| RTECS # | SZ3500000 |
運輸資料
| HS編碼* | 2931.49-900 |
Application
t-Butyldimethylsilylation of Alcohols using TBS Reagents

References
- 1)A New Method for Silylation of Hydroxylic Compounds: Reaction of Phenols and Alcohols with Silanols Mediated by Diethyl Azodicarboxylate and Triphenylphosphine
- 2)Protection for the Hydroxyl Group, Including 1,2- and 1,3-Diols
- P. G. M. Wuts, in Greene’s Protective Groups in Organic Synthesis, 5th ed., ed. by P. G. M. Wuts, John Wiley & Sons, Inc., Hoboken, New Jersey, 2014, Chap. 2, 17.
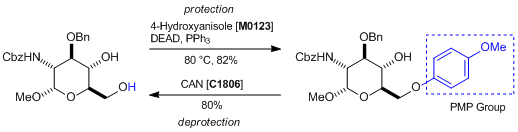
Application
Protection and deprotection of PMP groups
Reference
- Protection for the Hydroxyl Group, Including 1,2- and 1,3-Diols
- P. G. M. Wuts, in Greene’s Protective Groups in Organic Synthesis, 5th ed., ed. by P. G. M. Wuts, John Wiley & Sons, Inc., Hoboken, New Jersey, 2014, Chap. 2, 17.
Application
Corey-Fuchs Alkyne Synthesis

Typical Procedure:
Carbon tetrabromide (34.67 g, 104.5 mmol) and anhydrous CH2Cl2 (175 mL) are added to a 500 mL round bottom flask equipped with a magnetic stir bar and septum under N2 atmosphere. The reaction flask is cooled to 0 ℃ with an ice bath for 10 min. Triphenylphosphine (54.84 g, 209.1 mmol) is added in portions over 5 min, and the dark red solution is stirred at 0 ℃ for 30 min. 3,4-Dimethoxybenzaldehyde (17.37 g, 104.5 mmol) is added and the mixture is stirred at 0 ℃ for 1 h. A 1 : 1 mixture of H2O : brine is added and the layers are separated. The aqueous layer is extracted with a 1 : 1 mixture of hexanes : CH2Cl2, and the combined organic layers are dried, filtered, and concentrated under reduced pressure. The crude material is chromatographed on SiO2 (0-15% EtOAc/hexanes) to afford the dibromide 1 (29.37 g, Y. 88%) as a yellow oil.
Then, 1 (4.41 g, 13.79 mmol, 1.0 equiv) and THF (45 mL) are added to a 250 mL round bottom flask equipped with a magnetic stir bar and septum, and the reaction flask is cooled to -78 ℃ under N2 atmosphere. 2.5M n-BuLi solution in hexane (11.6 mL) is added slowly via syringe and the reaction solution is stirred at -78 ℃ for 45 min and then at 0 ℃ for 45 min. The flask is recooled to -78 ℃, and freshly distilled methyl chloroformate (1.1 mL, 1.3 mmol, 1.0 equiv) is added slowly via syringe. The mixture is stirred at -78 ℃ for 10 min, then 0 ℃ for 1 h. Saturated aqueous NH4Cl solution is added, and the layers are separated. The aqueous layer is extracted with Et2O, and the combined organic layers are dried, filtered and concentrated under reduced pressure. The residue is chromatographed (SiO2, 2-20% EtOAc / hexanes) to afford the desired alkyne 2 (2.83 g, Y. 93%) as a white solid.
Carbon tetrabromide (34.67 g, 104.5 mmol) and anhydrous CH2Cl2 (175 mL) are added to a 500 mL round bottom flask equipped with a magnetic stir bar and septum under N2 atmosphere. The reaction flask is cooled to 0 ℃ with an ice bath for 10 min. Triphenylphosphine (54.84 g, 209.1 mmol) is added in portions over 5 min, and the dark red solution is stirred at 0 ℃ for 30 min. 3,4-Dimethoxybenzaldehyde (17.37 g, 104.5 mmol) is added and the mixture is stirred at 0 ℃ for 1 h. A 1 : 1 mixture of H2O : brine is added and the layers are separated. The aqueous layer is extracted with a 1 : 1 mixture of hexanes : CH2Cl2, and the combined organic layers are dried, filtered, and concentrated under reduced pressure. The crude material is chromatographed on SiO2 (0-15% EtOAc/hexanes) to afford the dibromide 1 (29.37 g, Y. 88%) as a yellow oil.
Then, 1 (4.41 g, 13.79 mmol, 1.0 equiv) and THF (45 mL) are added to a 250 mL round bottom flask equipped with a magnetic stir bar and septum, and the reaction flask is cooled to -78 ℃ under N2 atmosphere. 2.5M n-BuLi solution in hexane (11.6 mL) is added slowly via syringe and the reaction solution is stirred at -78 ℃ for 45 min and then at 0 ℃ for 45 min. The flask is recooled to -78 ℃, and freshly distilled methyl chloroformate (1.1 mL, 1.3 mmol, 1.0 equiv) is added slowly via syringe. The mixture is stirred at -78 ℃ for 10 min, then 0 ℃ for 1 h. Saturated aqueous NH4Cl solution is added, and the layers are separated. The aqueous layer is extracted with Et2O, and the combined organic layers are dried, filtered and concentrated under reduced pressure. The residue is chromatographed (SiO2, 2-20% EtOAc / hexanes) to afford the desired alkyne 2 (2.83 g, Y. 93%) as a white solid.
References
- Total synthesis of (+)-Lithospermic acid by asymmetric intramolecular alkylation via catalytic C-H bond activation
Application
Corey-Nicolaou Macrolactonization1)

Typical Procedure:
To a stirred solution of the hydroxy carboxylic acid 1 (125 mg, 0.422 mmol) in dry CH2Cl2 (15 mL) is added triphenylphosphine (125 mg, 0.48 mmol) followed by 2,2’-dipyridyl disulfide (105 mg, 0.48 mmol) and the mixture is stirred until TLC (ethyl acetate : hexane = 2 : 3) shows complete loss of starting material (usually 0.5 h – 2 h). The mixture is concentrated in vacuo and dissolved in dry toluene (150 mL). To the solution is added 0.006M AgBF4 solution in toluene (1.2 mL) and the mixture is heated under reflux for 12 h. The mixture is cooled and solvents removed in vacuo. The residue is purified by flash column chromatography (acetone : CH2Cl2 = 0.5 : 99.5) to afford the desired lactone 2 (95 mg, Y. 81%).
To a stirred solution of the hydroxy carboxylic acid 1 (125 mg, 0.422 mmol) in dry CH2Cl2 (15 mL) is added triphenylphosphine (125 mg, 0.48 mmol) followed by 2,2’-dipyridyl disulfide (105 mg, 0.48 mmol) and the mixture is stirred until TLC (ethyl acetate : hexane = 2 : 3) shows complete loss of starting material (usually 0.5 h – 2 h). The mixture is concentrated in vacuo and dissolved in dry toluene (150 mL). To the solution is added 0.006M AgBF4 solution in toluene (1.2 mL) and the mixture is heated under reflux for 12 h. The mixture is cooled and solvents removed in vacuo. The residue is purified by flash column chromatography (acetone : CH2Cl2 = 0.5 : 99.5) to afford the desired lactone 2 (95 mg, Y. 81%).
References
- 1)Efficient and mild lactonization method for the synthesis of macrolides
- 2)Facile synthesis of saturated eight-membered ring lactones
考研文獻
文章/手冊
TCIMail
[TCIMAIL No.156] Mitsunobu Reaction Using Acetone Cyanohydrin[Product Highlights] Chiral Derivatization Reagent Possessing a Pyridylthiourea Structure
[TCI Practical Example] Bromination of an Alkyl Alcohol through the Appel Reaction
產品文件 (部分產品的分析圖譜無法提供,敬請諒解。)
SDS
請選擇語言。
請求的SDS不可用。
如需更多幫助,請聯繫我們 。
產品規格
檢驗報告(CoA)及其他文檔
請輸入批號
輸入的批號不正確
示例 CoA
可下載CoA示例。注:該示例可能非最新批次的CoA。
目前沒有該產品的 CoA 示例。
分析圖譜
請輸入批號
輸入的批號不正確
很抱歉,您搜索的分析圖譜無法提供。





