Maintenance Notice (3:15 - 7:30 AM February 22, 2025 UTC): This website is scheduled to be unavailable due to maintenance. We appreciate your patience and understanding.
Notice of Discontinuing the Use of Password-Protected Compressed Files | Product Document Searching Made Easy by 2D Code! | TCI Life Science News February 2025 | [Product Highlights] Chemiluminescent Reagents for... | Various analytical charts can be searched on each product detail page and Product Document Search (The kinds of analytical charts differ by product)
Maximum quantity allowed is 999
Please select the quantity
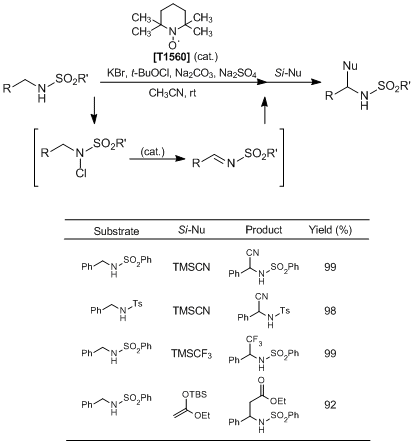
Nitroxyl-radical-catalyzed Oxidative Coupling of Amides with Silylated Nucleophiles
Moriyama et al. have reported the nitroxyl-radical-catalyzed oxidative coupling of amides with silylated nucleophiles. According to their results, in situ generated N-halogenated amides from amides and a halogenation reagent are activated by the treatment of a nitroxyl-radical TEMPO catalyst to afford the corresponding imine intermediates, which further react with silylated nucleophiles giving the desired coupling products in high yields. This reaction opened up new frontiers in nitroxyl-radical-catalyzed reactions, so it is expected that nitroxyl radicals can be used as organocatalysts in various transformations.