Maximum quantity allowed is 999
请选择数量
CAS RN: 247089-85-6 | 产品编码: T2814
(R)-(-)-p-Toluenesulfinamide

| 产品编码 | T2814 |
| 纯度/分析方法 | >98.0%(HPLC) |
| 分子式/分子量 | C__7H__9NOS = 155.22 |
| 外观与形状(20°C) | 固体 |
| 储存温度 | 室温 (15°C以下阴凉干燥处) |
| 包装和容器 | 1G-Glass Bottle with Plastic Insert (查看图片), 200MG-Glass Bottle with Plastic Insert (查看图片) |
| CAS RN | 247089-85-6 |
| Reaxys-RN | 8110323 |
| PubChem物质ID | 354334152 |
| MDL编号 | MFCD06858374 |
技术规格
| Appearance | White to Orange to Green powder to crystal |
| Purity(HPLC) | min. 98.0 area% |
| Purity(with Total Nitrogen) | min. 97.0 % |
| Melting point | 116.0 to 120.0 °C |
| Specific rotation [a]20/D | -81.0 to -91.0 deg(C=1, CHCL3) |
物性(参考值)
| 熔点 | 118 °C |
| 比旋光度 [α]D | -86° (C=1,CHCl3) |
| 溶解性(可溶于) | 甲醇 |
GHS
相关法规
运输信息
| HS编码* | 2930.90-900 |
应用
Sufinamide for the Synthesis of Chiral Amines
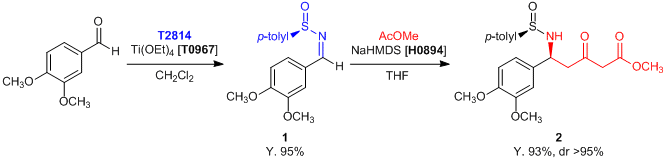
Experimental procedure (1 to 2): In a flask equipped with argon is placed a solution of THF (100 mL) and NaHMDS (1.0 mol/L solution in THF, 148 mL, 148 mmol, 6 eq.) at -78 °C. Methyl acetate (7.85 mL, 98.8 mmol, 4 eq.) in THF (20 mL) is added slowly, and the solution is stirred for 1 h. At this time anhydrous ether (20 mL) and 1 (7.48 g, 24.7 mmol, 1 eq.) in THF (20 mL) are added. The reaction mixture is stirred at -78 °C for 3.5 h and quenched with sat. NH4Cl (50 mL). The solution is washed with ethyl acetate (3 x 150 mL), the organic phases are combined, dried (Na2SO4), and concentrated. Purification by flash chromatography (50% EtOAc/hexane) gives 2 (8.80 g, 85% yield) as an oil.
References
- Alkaloid Synthesis Using Chiral δ-Amino β-Ketoesters: A Stereoselective Synthesis of (-)-Lasubine II
参考文献






