Maximum quantity allowed is 999
请选择数量
CAS RN: 2926-29-6 | 产品编码: T2033
Sodium Trifluoromethanesulfinate

技术规格
| Appearance | White to Orange to Green powder to crystal |
| Purity(Sodium hypochlorite Method) | min. 95.0 % |
物性(参考值)
| 水溶性 | 可溶 |
GHS
| 象形图 |

|
| 信号词 | Warning |
| 危险性说明 | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| 防范说明 | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P302 + P352 : IF ON SKIN: Wash with plenty of water. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P305 + P351 + P338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P362 + P364 : Take off contaminated clothing and wash it before reuse. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. |
相关法规
运输信息
| HS编码* | 2930.90-900 |
应用
Manganese-catalyzed Aerobic Oxytrifluoromethylation of Styrene Derivatives
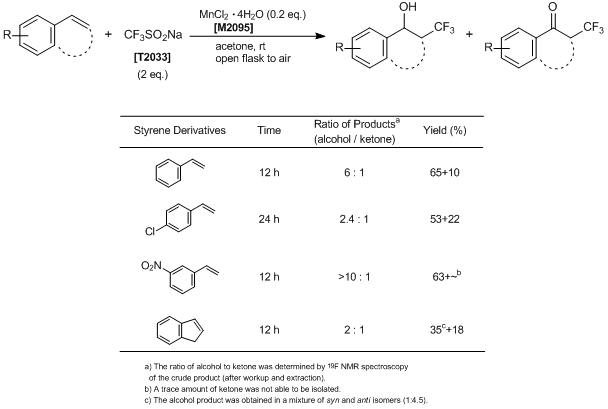
Typical Procedure:
To a solution of CF3SO2Na (128 mg, 0.8 mmol) in acetone (4 mL) are added styrenes (0.4 mmol) and then MnCl2·4H2O (16 mg, 0.08 mmol). The reaction mixture is stirred vigorously under an open atmosphere at room temperature for 12-48 h until the styrene substrate disappears by TLC monitoring. After completion of the reaction, the reaction mixture is poured into 5% aqueous NaHCO3 solution (20 mL) and then is diluted with ether (20 mL) and filtered through Celite. The filtrate is separated, and the aqueous layer is extracted by ether (20 mL). The organic phase is washed with saturated NaCl aqueous solution and then dried over sodium sulfate. After removal of the solvent in vacuo, the residue is purified by flash chromatography (hexane : ethyl acetate = 20 : 1 to 8 : 1) on silica gel to afford the corresponding ketone and alcohol products, respectively.
To a solution of CF3SO2Na (128 mg, 0.8 mmol) in acetone (4 mL) are added styrenes (0.4 mmol) and then MnCl2·4H2O (16 mg, 0.08 mmol). The reaction mixture is stirred vigorously under an open atmosphere at room temperature for 12-48 h until the styrene substrate disappears by TLC monitoring. After completion of the reaction, the reaction mixture is poured into 5% aqueous NaHCO3 solution (20 mL) and then is diluted with ether (20 mL) and filtered through Celite. The filtrate is separated, and the aqueous layer is extracted by ether (20 mL). The organic phase is washed with saturated NaCl aqueous solution and then dried over sodium sulfate. After removal of the solvent in vacuo, the residue is purified by flash chromatography (hexane : ethyl acetate = 20 : 1 to 8 : 1) on silica gel to afford the corresponding ketone and alcohol products, respectively.
References
应用
Radical Trifluoromethylation using Langlois Reagent

Reference
参考文献
文章/手册
产品文档 (部分产品的分析图谱无法提供,敬请谅解。)
化学品安全说明书(SDS)
请选择语言。
如需更多帮助,请联系我 们。
技术规格
CoA及其他文档
请输入批号
批号输入有误。请输入中横线前的4-5个字母数字字符。
示例 CoA
可下载CoA示例。注:该示例不一定是最新批次的CoA。
目前没有该产品的 CoA 示例。
分析图谱
请输入批号
批号输入有误。请输入中横线前的4-5个字母数字字符。
很抱歉,您搜索的分析图谱无法提供。






