Maintenance Notice (3:15 AM - 2:55 PM May 24, 2025 UTC): This website is scheduled to be unavailable due to maintenance. We appreciate your patience and understanding.
Product Document Searching Made Easy by 2D Code! | TCI Materials Science News May 2025 | [Product Highlights] Endogenous Biotin-Blocking Reagent... | Various analytical charts can be searched on each product detail page and Product Document Search (The kinds of analytical charts differ by product)
Maximum quantity allowed is 999
Please select the quantity
A Guanidinylating Reagent without Using Mercury Salts
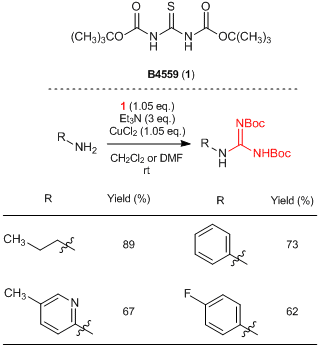
1,3-Bis(tert-butoxycarbonyl)thiourea(1) is a thiourea derivative and it can be used for the guanydinylation of amines. Generally, mercury salts are needed to effect a nucleophilic addition of amines to a thiocarbonyl moiety in thioureas. On the other hand, 1 can convert an amino group of primary amines into a guanidino group without using mercury salts, by the treatment of copper(II) chloride in the presence of triethylamine. This reaction proceeds under mild reaction conditions and various amines such as alkylamines and less nucleophilic aniline derivatives are susceptible to this transformation. It is known that some compounds such as tetrodotoxin and zanamivir have shown strong bioactivities. Thus, 1 is expected to be used as a strong tool for medicinal research because guanidinylations can be performed without using harmful mercury salts.
References
- Copper(II) chloride promoted transformation of amines into guanidines and asymmetrical N,N’-disubstituted guanidines