Make sure to sign up for an account today for exclusive coupons and free shipping on orders over $75!
Maximum quantity allowed is 999
CAS RN: 12092-47-6 | Product Number: B1045
Chloro(1,5-cyclooctadiene)rhodium(I) Dimer

Purity:
- Bis(1,5-cyclooctadiene)dirhodium(I) Dichloride
- 1,5-Cyclooctadiene Rhodium(I) Chloride Dimer
- [Rh(COD)Cl]2
| Size | Unit Price | Philadelphia, PA | Portland, OR | Japan* | Quantity |
|---|---|---|---|---|---|
| 100MG |
$89.00
|
2 | 2 | 33 |
|
| 1G |
$523.00
|
9 | 3 | ≥100 |
|
* Items in stock locally ship in 1-2 business days. Items from Japan stock are able to ship from a US warehouse within 2 weeks. Please contact TCI for lead times on items not in stock. Excludes regulated items and items that ship on ice.
* To send your quote request for bulk quantities, please click on the "Request Quote" button. Please note that we cannot offer bulk quantities for some products.
*TCI frequently reviews storage conditions to optimize them. Please note that the latest information on the storage temperature for the products is described on our website.
| Product Number | B1045 |
| Molecular Formula / Molecular Weight | C__1__6H__2__4Cl__2Rh__2 = 493.08 |
| Physical State (20 deg.C) | Solid |
| Storage Temperature | Room Temperature (Recommended in a cool and dark place, <15°C) |
| Packaging and Container | 100MG-Glass Bottle with Plastic Insert (View image), 1G-Glass Bottle with Plastic Insert (View image) |
| CAS RN | 12092-47-6 |
| Reaxys Registry Number | 14860700 |
| PubChem Substance ID | 87563994 |
| MDL Number | MFCD00012415 |
| Appearance | Light yellow to Brown powder to crystal |
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P305 + P351 + P338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P302 + P352 : IF ON SKIN: Wash with plenty of soap and water. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. P362 : Take off contaminated clothing and wash before reuse. |
| HS Number | 2931.90.9052 |
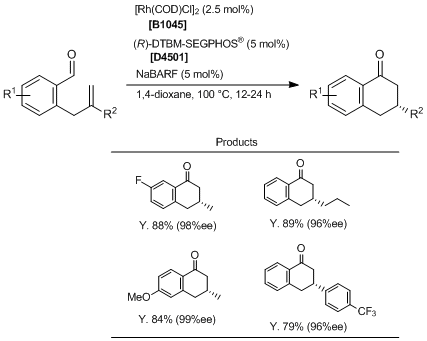
In a nitrogen-filled dry box, the appropriate o-allylbenzaldehyde (0.20 mmol, 1.0 eq.), [Rh(COD)Cl]2 (2.5 mg, 0.0050 mmol, 0.025 eq.), (R)-DTBM-SEGPHOS® (11.8 mg, 0.010 mmol, 0.050 eq.), NaBARF (8.9 mg, 0.010 mmol, 0.050 eq.), and anhydrous 1,4-dioxane (1 mL) are added to a 1-dram vial. The vial is sealed with a PFTE/silicone-lined septum cap and removed from the dry box. The reaction mixture is heated to 100 °C and allowed to stir at this temperature until the reaction is judged to be complete by TLC analysis. The mixture is cooled to room temperature, filtered through a pad of silica gel (eluting with EtOAc), and concentrated under reduced pressure. The crude reaction mixture is purified by flash column silica gel chromatography (hexane/EtOAc or hexane/Et2O) to yield the corresponding products.
References

References

References
Articles/Brochures
Safety Data Sheet (SDS)
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Sample C of A
A sample C of A for this product is not available at this time.
Analytical Charts
The requested analytical chart is not available. Sorry for the inconvenience.





