Maximum quantity allowed is 999
CAS RN: 53199-31-8 | 제품번호: B3161
Bis(tri-tert-butylphosphine)palladium(0)
순도/분석 방법: >98.0%(T)
•본건의 원가격은 한국 대리점의 예상 판매가격입니다.자세한 정보가 필요하시면 연락해 주십시오.( SEJIN CI Co., Ltd. (한국총대리점) 전화 : 02-2655-2480 이메일 : sales@sejinci.co.kr)
•보관 조건은 예고없이 변경 될 수 있습니다. 제품 보관 조건의 최신 자료는 홈페이지에 기재되어 있으니 양해 부탁드립니다.
| 제품번호 | B3161 |
Purity/Analysis Method 
|
>98.0%(T) |
| M.F. / M.W. | C__2__4H__5__4P__2Pd = 511.06 |
| 물리적 상태 (20 ℃) | Solid |
보관 조건 
|
Frozen (<0°C) |
| 불활성 가스 하에서 보관 | Store under inert gas |
| 피해야 할 조건 | Air Sensitive,Heat Sensitive |
용기 
|
1G-Glass Bottle with Plastic Insert (이미지 보기) |
| CAS RN | 53199-31-8 |
| Reaxys-RN | 14300595 |
| PubChem Substance ID | 87560386 |
| MDL 번호 | MFCD03094580 |
| Appearance | White to Yellow to Orange powder to crystal |
| Purity(Chelometric Titration) | min. 98.0 % |
| HS 번호* | 2843.90-000 |
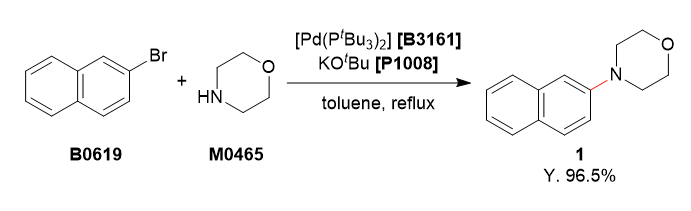
Used Chemicals
Procedure
To a 4-necked 200 mL flask was charged with 2-bromonaphthalene (5.20 g, 25.1 mmol, 1.0 equiv.), morpholine (3.27 mL, 37.5 mmol, 1.5 equiv.) and degassed toluene (75 mL). To this solution was added bis(tri-tert-butylphosphine)palladium(0) (256 mg, 0.501 mmol, 2.0 mol%) and potassium tert-butoxide (4.21 g, 37.5 mmol, 1.5 equiv.). The reaction mixture was refluxed for 3 h under argon atmosphere. The reaction mixture was cooled to room temperature and washed with water (50 mL). The organic layer was separated and the aqueous layer was extracted with ethyl acetate (50 mL). The combined organic layers were washed with brine (50 mL), dried over Na2SO4 and filtered. The solvent was removed under reduced pressure. The residue was purified by column chromatography (eluent: hexane/ethyl acetate, 85/15→70/30) to obtain 1 as a white solid (5.17 g, 96.5%).
Experimenter's Comments
The reaction mixture was monitored by TLC (hexane/ethyl acetate = 9/1, Rf = 0.70).
Analytical Data(Compound 1)
1H NMR (400 MHz, CDCl3); δ 7.76–7.69 (m, 3H), 7.42 (t, J = 7.2 Hz, 1H), 7.31 (t, J = 6.8 Hz, 1H), 7.25–7.28 (m, 1H), 7.13 (s, 1H), 3.92 (t, J = 4.8 Hz, 2H), 3.27 (t, J = 4.8 Hz, 2H).
13C NMR (101 MHz, CDCl3); δ 129.0, 127.6, 127.0, 126.5, 123.7, 119.1, 110.3, 67.1, 50.0.
Lead Reference
- Heterogeneous Rhodium‐Catalyzed Aerobic Oxidative Dehydrogenative Cross‐Coupling: Nonsymmetrical Biaryl Amines
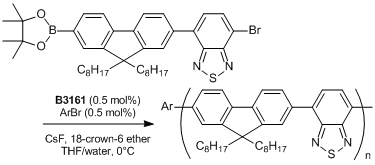
References
- Chain-Growth Suzuki Polymerization of n-Type Fluorene Copolymers
- E. Elmalem, A. Kiriy, W. T. S. Huck, Macromolecules 2011, 44, 9057.


Typical Procedure: An ampoule is charged with the thiocarbamate (0.442 mmol) and anhydrous degassed toluene (4 mL) is added via gastight syringe under nitrogen with stirring. A J. Youngs valve is fitted and the ampoule is placed into an oil bath pre-equilibrated at 100℃. After ca. 5 minutes the valve is removed and Pd(t-Bu3P)2 (2 mol%, 0.00884 mmol) is added as a solid, the valve is then replaced and the reaction mixture heated for 2.5 hours. A sample of the reaction mixture is removed via gastight syringe and found to contain the desired product (>99%).
Reference
- The Newman–Kwart Rearrangement of O‐Aryl Thiocarbamates: Substantial Reduction in Reaction Temperatures through Palladium Catalysis
문서
SDS
요청한 SDS를 사용할 수 없습니다.
번거롭게 해드려 죄송하지만 이 양식 보다 문의 해주십시오.
규격표
시험성적서, 각종 증명서
샘플 시험성적서
본 제품의 샘플시험성적서는 현재 준비되어 있지 않습니다.
분석 차트

죄송합니다만 찾으시는 분석차트는 없습니다.





