Maximum quantity allowed is 999
Please select the quantity
CAS RN: 1264520-63-9 | Product Number: T2937
O-TBDPS-D-Thr-N-Boc-L-tert-Leu-Diphenylphosphine
Purity: >98.0%(HPLC)
Synonyms:
- N-(tert-Butoxycarbonyl)-L-tert-leucine N-[(2R,3S)-3-(tert-Butyldiphenylsilyloxy)-1-(diphenylphosphino)-2-butyl]amide
- N-Boc-L-tert-leucine N-[(2R,3S)-3-(tert-Butyldiphenylsilyloxy)-1-(diphenylphosphino)-2-butyl]amide
Product Documents:
* Please contact our distributor of SEJIN CI Co., Ltd.
if you would like to purchase TCI products. The above prices do not include freight cost, customs charge and other charges to the destination. SEJIN CI Co., Ltd. (Phone: 02-2655-2480 / email:
sales@sejinci.co.kr)
* The storage conditions are subject to change without notice.
* The storage conditions are subject to change without notice.
| Product Number | T2937 |
| Purity / Analysis Method | >98.0%(HPLC) |
| Molecular Formula / Molecular Weight | C43H57N2O4PSi = 725.00 |
| Physical State (20 deg.C) | Solid |
| Storage Temperature | Room Temperature (Recommended in a cool and dark place, <15°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Air Sensitive |
| Packaging and Container | 100MG-Glass Bottle with Plastic Insert (View image) |
| CAS RN | 1264520-63-9 |
| Reaxys Registry Number | 21221495 |
| PubChem Substance ID | 172089008 |
Articles/Brochures
TCIMAIL
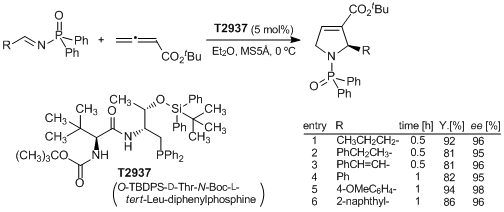
[TCIMAIL No.158] A Novel Dipeptide-Based Chiral Phosphine Organocatalyst for Asymmetric Cyclization[Research Articles] A highly Enantioselective [3+2] Cyclization between Imines and Allenoates Catalyzed by a Dipeptide-Based Chiral Phosphine
Product Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.