Published TCIMAIL newest issue No.197
Maximum quantity allowed is 999
CAS RN: 133745-75-2 | Produkte #: F0335
N-Fluorobenzenesulfonimide [Fluorinating Reagent]
![N-Fluorobenzenesulfonimide [Fluorinating Reagent] No-Image](/medias/F0335.jpg?context=bWFzdGVyfHJvb3R8NDYzMDV8aW1hZ2UvanBlZ3xhREZrTDJnMFlTODRPVEl4TmpnNU16VTRNelkyTDBZd016TTFMbXB3Wnd8NDJkMWVkY2MxYjM2N2FjOWNlNDk2ZjkxOWJhNjljYWEwNmQzMzY5ZTBhYWFlMDYyOGMxM2M5NmIzNjA3MmVhOQ)
Reinheit: >98.0%(T)(HPLC)
- N-Fluorobis(phenylsulfonyl)amine
- NFSI
- NFBS
| Einheit | Stückpreis | Belgien | Japan* | Menge |
|---|---|---|---|---|
| 5G |
€42.00
|
5 | ≥100 |
|
| 25G |
€163.00
|
3 | ≥100 |
|
*In Belgien verfügbare Lagerbestände werden in 1 bis 3 Tagen geliefert.
*In Japan verfügbare Lagerbestände werden in 1 bis 2 Wochen geliefert. (unter Ausschluss von regulierten Artikeln und Trockeneislieferungen).
| Artikel # | F0335 |
| Reinheit / Analysenmethode | >98.0%(T)(HPLC) |
| Summenformel / Molekülmasse | C__1__2H__1__0FNO__4S__2 = 315.33 |
| Physikalischer Zustand (20 °C) | Solid |
| Lagerungstemperatur | Room Temperature (Recommended in a cool and dark place, <15°C) |
| CAS RN | 133745-75-2 |
| Reaxys Registrierungsnummer | 5348902 |
| PubChem-Stoff-ID | 87570107 |
| Spektrale Daten (AIST) Link | 19601 |
| MDL-Nummer | MFCD00144885 |
| Appearance | White to Light yellow powder to crystal |
| Purity(HPLC) | min. 98.0 area% |
| Purity(Iodometric Titration) | min. 98.0 % |
| Melting point | 115.0 to 119.0 °C |
| Schmelzpunkt | 115 °C |
| Piktogramm |


|
| Signalwort | Achtung |
| Gefahrenhinweise | H315 : Verursacht Hautreizungen. H319 : Verursacht schwere Augenreizung. H341 : Kann vermutlich genetische Defekte verursachen. |
| Sicherheitshinweise | P501 : Inhalt/ Behälter einer anerkannten Abfallentsorgungsanlage zuführen. P201 : Vor Gebrauch besondere Anweisungen einholen. P264 : Nach Gebrauch Haut gründlich waschen. P280 : Schutzhandschuhe/ Schutzkleidung/ Augenschutz/ Gesichtsschutz/ Gehörschutz Tragen. P308 + P313 : BEI Exposition oder falls betroffen: Ärztlichen Rat einholen/ ärztliche Hilfe hinzuziehen. P337 + P313 : Bei anhaltender Augenreizung: Ärztlichen Rat einholen/ ärztliche Hilfe hinzuziehen. |
| HS-Nr. (Import / Export) (TCI-E) | 2935909099 |

-
Used Chemicals
-
Procedure
-
To a solution of 4-bromobiphenyl (1.17 g, 5.00 mmol), DABSO (721 mg, 3.00 mmol), and PdCl2(Amphos)2 (177 mg, 0.25 mmol) in anhydrous isopropyl alcohol (20 mL) was added triethylamine (2.1 mL, 15 mmol) at room temperature and the mixture was stirred at 75 °C for 23 hours. The suspension was cooled at room temperature and NFSI (2.36 g, 7.5 mmol) was added and the reaction mixture stirred for 3 hours until completion. The solvent was removed under reduced pressure. The residue was diluted with ethyl acetate (20 mL) and filtrated through celite pad. The filtrate was washed with saturated aqueous Na2S2O3 solution (20 mL) and brine (20 mL), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by medium pressure preparative chromatography (ethyl acetate:hexane = 2:98 - 5:95 on silica gel) to give compound 1 as a white solid (928 mg, 79%).
-
Experimenter’s Comments
-
The reaction mixture was monitored by TLC (ethyl acetate:hexane = 1:9, Rf = 0.57) and UPLC-MS.
-
Analytical Data
-
Compound 1
1H NMR (400 MHz, CDCl3); δ 8.12–8.04 (m, 2H), 7.86–7.80 (m, 2H), 7.67–7.58 (m, 2H), 7.44-7.55 (m, 3H).
-
Lead Reference
-
- One-pot palladium-catalyzed synthesis of sulfonyl fluorides from aryl bromides
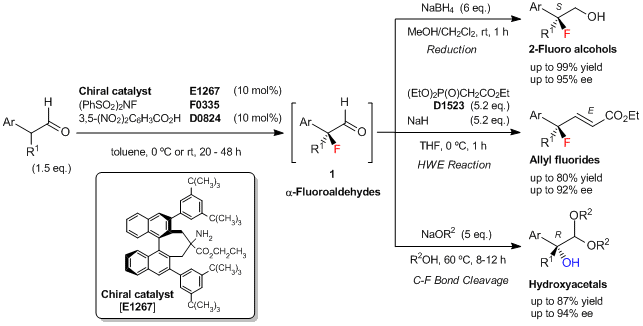
To a solution of the chiral amine catalyst [E1267] (20 mg, 0.026 mmol, 10 mol%) in toluene (0.54 mL) is added 3,5-dinitrobenzoic acid (5.5 mg, 0.026 mmol, 10 mol%), 2-phenylpropionaldehyde (52 mg, 0.39 mmol, 1.5 eq.), and N-fluorobenzenesulfonimide (NFSI) (0.26 mmol, 82 mg, 1 eq.) at 0 °C. The reaction mixture is stirred at 0 °C for 48 h, then poured into CH3OH/CH2Cl2 (1:4, 1.3 mL) at 0 °C. To this solution, NaBH4 (1.6 mmol, 6 eq.) is added, and the mixture is stirred at room temperature for 1 h. The reaction is quenched with saturated aq.NH4Cl, and the mixture is extracted with Et2O. The organic layer is dried over Na2SO4, and concentrated. The residue is purified by silica gel chromatography (eluent: hexane/ethyl acetate = 3/1) to give (S)-2-fluoro-2-phenylpropan-1-ol (34.5 mg, 86% yield based on NFSI, 95% ee) as a white solid.
References

In a dry 25 mL round-bottom flask, thiophene (0.5 mmol, 1 eq.), NFSI (0.6 mmol, 1.2 eq.) and CuI (5 mol%) are dissolved in DCE (3 mL), the resulting mixture is put into a preheated oil bath (60 °C) for 8 h. The solution is then cooled to room temperature, evaporated under reduced pressure, diluted with ethyl acetate, washed with a saturated aqueous solution of NaCl, dried over Na2SO4, filtered and evaporated under reduced pressure. The crude product is purified by silica gel column chromatography to afford the corresponding product (166.7 mg, Y. 88 %).
References

Reference
Artikel / Broschüren
[Research Articles] Copper-catalyzed Direct Amidation of Heterocycles with N-Fluorobenzenesulfonimide
[TCI Practical Example] One-Pot Palladium-Catalyzed Fluorosulfonylation of an Aryl Bromide Using DABSO and NFSI
Sicherheitsdatenblatt (SDB)
Das angeforderte SDB ist nicht verfügbar.
Bitte Kontaktieren Sie uns für mehr Informationen.
Spezifikationsdokumenten
AZ & andere Zertifikate
Muster-AZ
Ein Muster-AZ für dieses Produkt ist zur Zeit nicht verfügbar.
Analytische Diagramme
Das angeforderte Analysediagramm ist nicht verfügbar. Wir entschuldigen uns für die Unannehmlichkeiten.



![N-Fluorobenzenesulfonimide [Fluorinating Reagent] N-Fluorobenzenesulfonimide [Fluorinating Reagent]](/_ui/responsive/theme-tci/images/missing_product_zoom.jpg)

